Obicetrapib: Dual Cardiovascular and Alzheimer's Benefit Potential
This deep dive into obicetrapib, an investigational drug, reveals a complex interplay between lipid metabolism, cardiovascular health, and potential neurodegenerative disease prevention, particularly for APOE4 carriers. The conversation unpacks the checkered history of CETP inhibitors, highlighting how early failures stemmed from focusing solely on raising HDL or from significant off-target toxicities. Obicetrapib's promise lies in its robust LDL and ApoB reduction without adverse effects, coupled with intriguing biomarker shifts in Alzheimer's-related markers. This analysis is crucial for anyone involved in cardiovascular disease management or exploring novel avenues for neurodegenerative disease prevention, offering a glimpse into how a drug initially developed for heart health might offer unexpected benefits for brain health, especially for genetically predisposed individuals. The hidden consequence of previous CETP inhibitor failures was the missed opportunity for a more nuanced approach to lipid management; obicetrapib may rectify this by demonstrating a more sophisticated understanding of lipid pathways.
The Unforeseen Cascade: From Lipid Ripples to Brain Waves
The journey of obicetrapib from a class of drugs with a troubled past to a beacon of cautious optimism is a masterclass in scientific iteration and understanding downstream effects. Early attempts to harness the power of CETP inhibition were largely derailed by a simplistic view of HDL cholesterol and, in some cases, catastrophic off-target toxicities. This history underscores a critical lesson: interventions targeting complex biological systems rarely have a single, predictable outcome. Obicetrapib's potential success hinges on its ability to navigate these complexities, offering not just cardiovascular benefits but also intriguing signals for Alzheimer's disease, particularly in APOE4 carriers.
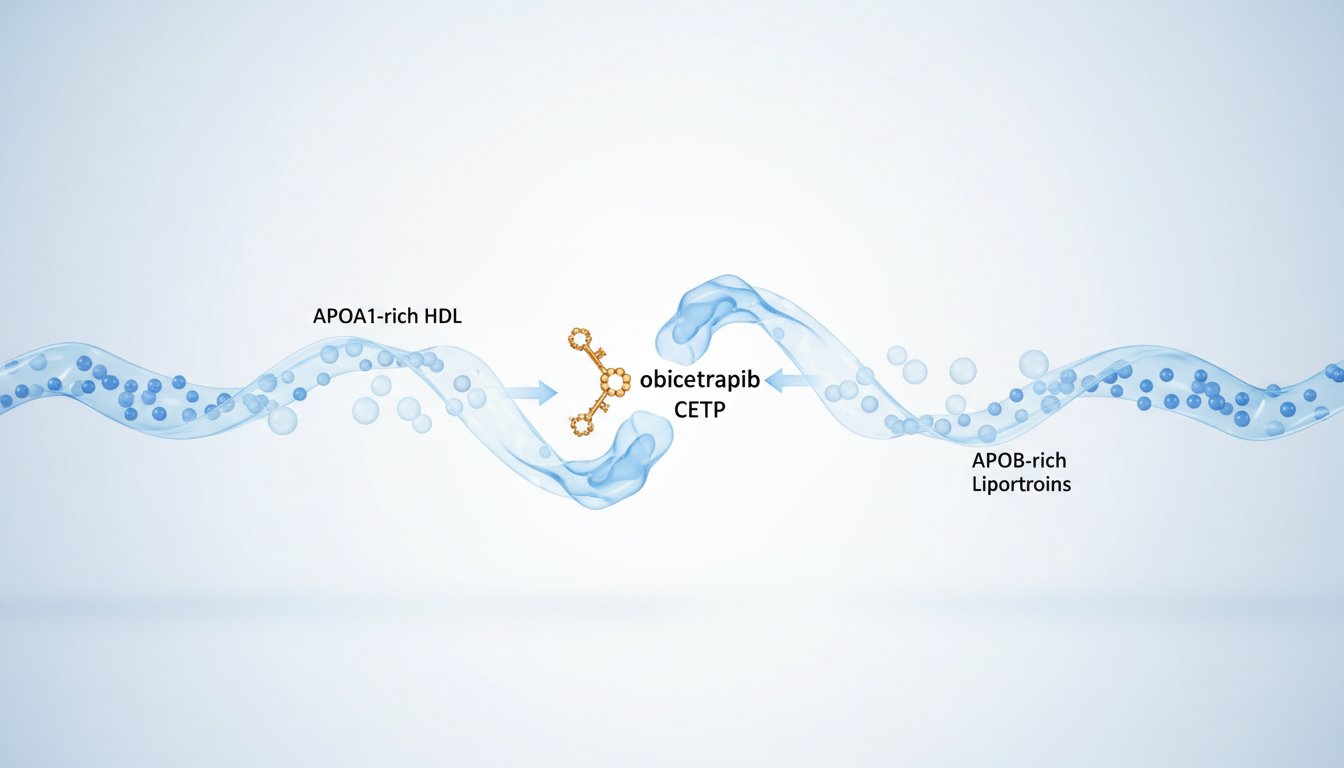
The core of this story lies in understanding reverse cholesterol transport and the intricate dance of lipoproteins. Lipoproteins, essential for moving water-insoluble cholesterol through the bloodstream, are broadly categorized into ApoB (like LDL) and ApoA1 (like HDL) classes. While ApoB particles are known culprits in atherosclerosis, HDLs, mediated by ApoA1, have historically been viewed as beneficial, carrying cholesterol back to the liver. However, the reality is far more nuanced.
"The direct RCT or reverse cholesterol transport is when the HDL delivers that cholesterol straight into the liver... But there's also something called indirect RCT. And I don't think I even learned what indirect RCT was until maybe eight or nine years ago..."
-- Peter Attia
This indirect pathway, where HDL exchanges cholesterol esters for triglycerides in ApoB particles, highlights that LDLs aren't entirely detrimental; they play a role in returning cholesterol to the liver. CETP (cholesterol ester transfer protein) acts as a shuttle in this exchange. High CETP activity leads to cholesterol-poor, triglyceride-rich HDLs and cholesterol-rich, triglyceride-poor LDLs. While this might seem beneficial for HDL, the cholesterol-rich LDLs can still contribute to atherosclerosis. Inhibiting CETP, conversely, results in larger, cholesterol-rich HDLs and lower LDL cholesterol.
The early CETP inhibitors, developed in the 1990s, focused heavily on the HDL-raising aspect. The prevailing wisdom was that "HDL is good cholesterol," so raising it should be beneficial. This proved to be an overly simplistic assumption. Mendelian randomization studies later failed to support a causal link between simply raising HDL cholesterol and favorable cardiovascular outcomes, starkly contrasting with the consistent findings for LDL cholesterol. Furthermore, genetic insights revealed that individuals with loss-of-function mutations in the HDL receptor SR-B1, despite having very high HDL cholesterol, exhibited increased coronary artery disease risk. This complex biology meant that simply increasing HDL wasn't enough; the quality and function of the particles, along with LDL reduction, were critical.
The graveyard of early CETP inhibitors is littered with cautionary tales. Torcetrapib was withdrawn due to increased mortality from off-target blood pressure effects. Dalcetrapib, while safe, failed to show meaningful cardiovascular benefits because it didn't adequately lower LDL or ApoB. Evacetrapib showed modest LDL reduction but was ultimately abandoned. Anacetrapib demonstrated a modest cardiovascular benefit, but Merck ultimately pulled it, possibly due to a long half-life and fat cell accumulation, despite no proven harm. These failures, while disheartening, paved the way for a more refined understanding, leading to obicetrapib.
"So you go, what is that, five drugs, or four drugs that go O for four, or at least three of them go O for three, and maybe the fourth one kind of hits, but has this weird issue of getting held up in fat cells, and therefore they decide, forget it, we're not going to take that risk."
-- Peter Attia
Obicetrapib's distinct advantage lies in its robust LDL and ApoB reduction--an additional 50% reduction on top of high-intensity statin therapy in the ROSE trial. It also demonstrated a significant 30% reduction in LDL in the BROADWAY trial, alongside a notable 16% decrease in ApoB. Crucially, it appears to be metabolically neutral or even beneficial, unlike statins which can increase the risk of type 2 diabetes. This comprehensive lipid-modifying profile, including a one-third reduction in Lp(a), a significant independent cardiovascular risk factor, positions obicetrapib as a potentially transformative therapy for cardiovascular disease.
However, the most unexpected and exciting development comes from the BROADWAY trial's biomarker substudy. The brain, a lipid-rich organ, operates with a semi-independent lipid economy behind the blood-brain barrier, utilizing APOE for cholesterol transport. APOE4 carriers, particularly APOE4/4 homozygotes, have less efficient lipid transport in the brain, contributing to increased Alzheimer's risk. Obicetrapib, by potentially increasing functional ApoA1 availability within the central nervous system and augmenting cholesterol efflux, may offer a compensatory mechanism. The BROADWAY biomarker study showed that obicetrapib significantly attenuated the increase in p-tau 217, a key Alzheimer's biomarker, over 12 months. The effect was most pronounced in APOE4/4 individuals, where a remarkable 20% difference in p-tau 217 reduction was observed compared to placebo.
"But the most interesting finding for me, and I think anybody who would look at the paper, is what happened in the admittedly small subset, 29 people, of E4/E4s of any age."
-- Peter Attia
While this is a biomarker study and not a definitive outcome trial, the genotype specificity and biological plausibility are compelling. The implication is that a drug designed to manage cardiovascular risk might also offer a novel pathway for mitigating Alzheimer's disease pathology, turning a potential cardiovascular intervention into a dual-purpose therapeutic. This suggests that a deeper understanding of lipid dynamics could unlock unexpected therapeutic avenues, rewarding the patience required to explore complex biological systems.
Key Action Items
-
Immediate Action (Within the next quarter):
- For Healthcare Professionals: Review the available data on obicetrapib, focusing on its cardiovascular and Alzheimer's biomarker findings, particularly in APOE4 carriers. Understand its mechanism of action and historical context of CETP inhibitors.
- For Patients (especially APOE4 carriers at high cardiovascular risk): Discuss with your physician the potential role of emerging lipid-modifying therapies like obicetrapib, understanding that it is still investigational in many regions.
- For Researchers: Design prospective, long-term prevention trials specifically enriched for APOE4 carriers to confirm cognitive and imaging outcomes, moving beyond biomarker studies.
-
Short-Term Investment (Next 6-12 months):
- For Pharmaceutical Companies: Continue rigorous clinical trials for obicetrapib, focusing on cardiovascular outcomes (PREVAIL trial) and dedicated Alzheimer's prevention studies with sensitive cognitive endpoints.
- For Clinicians: Stay abreast of regulatory approvals for obicetrapib in your region, preparing for its potential integration into treatment protocols for residual cardiovascular risk.
-
Mid-Term Investment (12-18 months):
- For Patients: If obicetrapib becomes available, engage in shared decision-making with your doctor about its use, weighing its benefits for cardiovascular health and potential neurological protection against any known risks.
- For Researchers: Investigate the precise mechanisms by which obicetrapib influences brain lipid metabolism and its interaction with APOE isoforms, seeking to understand the "why" behind the observed biomarker shifts.
-
Longer-Term Strategic Focus (18+ months):
- For Public Health & Policy Makers: Consider the implications of therapies that offer dual cardiovascular and neurodegenerative benefits, potentially impacting long-term healthcare resource allocation and preventative strategies.
- For Individuals: Prioritize a holistic approach to health that includes understanding genetic predispositions (like APOE status) and engaging with emerging scientific advancements that offer potential for extended healthspan.
- For Drug Developers: Explore other CETP inhibitors or novel lipid-modulating agents that may offer similar or improved cardiovascular and neurological benefits, learning from obicetrapib's trajectory.